New Scripps Research Study: Rosemary Compound Restores Memory and Mitigates Alzheimer’s
Scripps researchers show that diAcCA, a rosemary-derived compound, can mitigate two hallmarks of Alzheimer’s and improve spatial memory and learning.
Highlights
- diAcCA reverses memory deficits and reduces hallmark Alzheimer’s pathology in a mouse model of Alzheimer’s disease.
- diAcCA, a derivative of carnosic acid, absorbs more efficiently and has a longer half-life, leading to greater brain bioavailability and sustained therapeutic exposure.
- The cognitive benefits of diAcCA are likely linked to the activation of the Nrf2 antioxidant and anti-inflammatory pathway.
Alzheimer’s disease (AD) is one of the most devastating age-related neurodegenerative conditions, marked by progressive memory loss, cognitive impairment, and widespread neuronal damage. It affects more than 50 million people worldwide and represents a major challenge for healthcare systems and aging populations. Despite extensive research, most available treatments provide only symptomatic relief, failing to address the underlying causes of the disease.
Emerging research suggests that targeting the brain’s natural defense mechanisms against oxidative stress and inflammation may offer new therapeutic opportunities. Moreover, a recent study published in Antioxidants highlights the potential of diAcCA, a novel pro-drug derived from carnosic acid found in rosemary, as a promising candidate for Alzheimer’s therapy. The study demonstrated that diAcCA restores synaptic health, reduces hallmark Alzheimer’s pathology, and improves cognitive function in a widely used mouse model of the disease.
Activating the Brain’s Antioxidant Pathway with diAcCA
The brain is highly susceptible to oxidative stress and chronic inflammation, which are key drivers of neuronal loss and cognitive decline in Alzheimer’s disease. A critical molecular pathway responsible for regulating the brain’s response to these insults is the Nrf2 pathway. When activated, Nrf2 controls the production of antioxidant and anti-inflammatory proteins, helping to protect cells from damage. However, in Alzheimer’s disease, Nrf2 activity is often suppressed, leaving neurons vulnerable to degeneration.
Carnosic acid (CA), a naturally occurring compound in rosemary and sage, has been shown to activate Nrf2 signaling. Unfortunately, its clinical potential has been limited by its chemical instability. CA rapidly degrades when exposed to air or light, resulting in poor shelf life and low bioavailability when administered orally.
In an effort to overcome these challenges, researchers at The Scripps Research Institute developed diAcCA, a drug derivative of CA that remains stable in storage and converts to active CA in the acidic environment of the stomach, enabling more consistent delivery of the therapeutic agent to the brain.
diAcCA Reduces Alzheimer’s Pathology in Mice
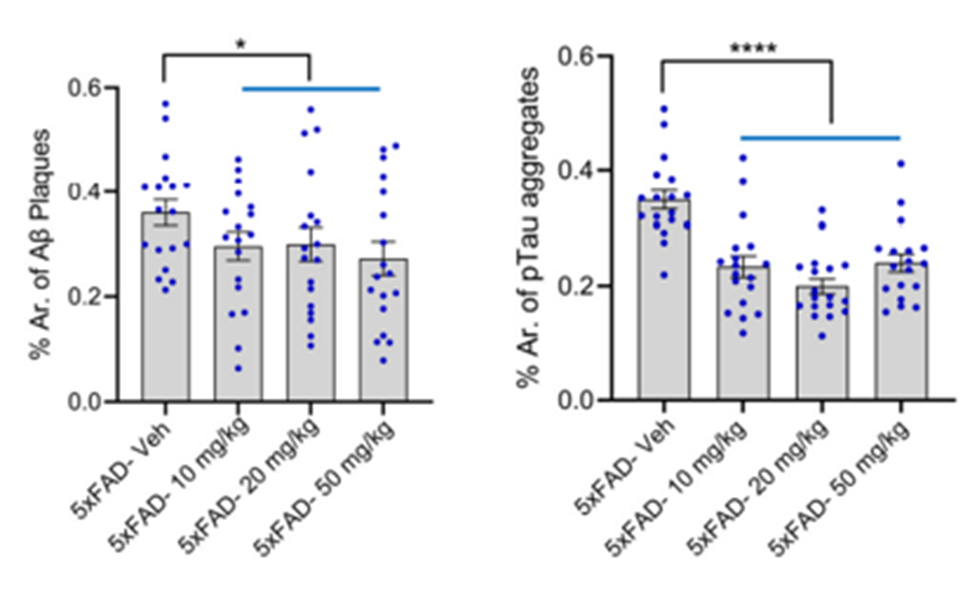
To see if diAcCA could be helpful as a treatment, the scientists tested it on mice that model Alzheimer’s called 5xFAD mice. These mice were genetically manipulated to harbor five human genes associated with Alzheimer’s that cause them to develop Alzheimer’s disease very quickly. Because of these genetic modifications, the mice show many of the same problems that people with Alzheimer’s have. This includes sticky clumps in the brain called amyloid plaques, twisted tangles of a protein called phosphorylated tau, loss of connections between neurons (called synapses), and serious memory problems. Scientists often use this model because it helps study Alzheimer’s disease in a way that’s similar to what happens in humans.
Mice were given diAcCA by mouth, using a tiny feeding tube, three times per week for three months. Notably, treatment began when mice were five months old, which is when cognitive deficits become apparent. By the end of the treatment period, the diAcCA-treated mice displayed significantly fewer amyloid-beta plaques and phosphorylated tau aggregates than untreated mice in the hippocampus, the part of the brain that helps with learning and memory.
In addition to lowering these harmful protein buildups, the diAcCA treatment also helped restore the number of healthy connections between neurons, known as synapses. The researchers found increased levels of two key proteins: Synapsin I, which indicates improved synapse function, and NeuN, a marker of healthy neurons. They also observed reduced activity in brain support cells called astrocytes and microglia, suggesting that diAcCA helped lower neuroinflammation, which is a major contributor to brain damage in Alzheimer’s disease.
diAcCA Improves Memory and Learning
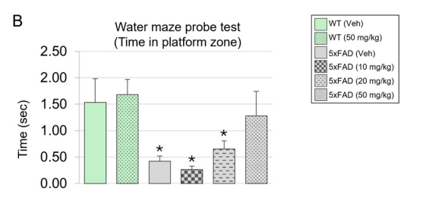
The brain improvements seen in the mice treated with diAcCA also led to better memory and learning. To test this, the researchers used something called the Morris water maze—a test where mice have to swim around a small pool and try to remember where a hidden platform is so they can climb out. Remarkably, the mice that received diAcCA were better at remembering where the platform was. They not only spent more time searching in the right area of the pool, but they also found the platform more quickly than untreated mice. This showed that their spatial memory—the ability to remember places and directions—had improved.
The researchers also ran another test called contextual fear conditioning, which checks if mice remember things by association. In this test, the mice learned to associate a certain place with an unpleasant experience. Interestingly, the treated mice showed stronger freezing behavior (they stayed very still), which means they remembered that the place was connected to something they didn’t like. This kind of memory also relies on the hippocampus. The fact that these mice did better suggests that diAcCA helped their hippocampus work more like it should, potentially reversing some of the damage caused by Alzheimer’s disease.
Safety and Stability: Addressing the Shortcomings of Carnosic Acid
A major strength of diAcCA lies in its ability to overcome the limitations that have long held back carnosic acid as a therapeutic agent. In preclinical studies, diAcCA exhibited improved behavior in the body—what researchers call pharmacokinetic properties, referring to how a compound is absorbed, distributed, metabolized, and excreted. Compared to carnosic acid alone, diAcCA showed approximately 20% greater bioavailability, meaning more of the compound successfully entered the bloodstream after administration. It also reached peak blood levels about 30% higher, suggesting more effective delivery of the active ingredient to target tissues.
In addition to enhanced absorption, diAcCA remained in the body longer, with a half-life exceeding 12 hours. A compound’s half-life refers to the amount of time it takes for half of the drug to be eliminated from the system—longer half-lives typically mean more stable and sustained therapeutic effects, allowing for less frequent dosing.
From a practical standpoint, diAcCA also offers greater chemical stability. It retained its structural integrity for over two years in storage, a marked improvement over the short shelf life of carnosic acid. This enhanced stability could help streamline manufacturing and storage processes in a clinical setting.
Importantly, toxicology assessments in mice showed that diAcCA was well-tolerated, even at higher doses, and caused no observable side effects. Since its active form—carnosic acid—is already classified by the U.S. Food and Drug Administration as Generally Recognized As Safe (GRAS), diAcCA emerges as a promising candidate for translation into human clinical trials.
Rethinking Neurodegeneration: Restoring Resilience in the Aging Brain
Although the findings remain limited to animal models, they call attention to a pathway—Nrf2 signaling—that may carry broader importance than previously appreciated. If further studies uphold the ability of diAcCA to preserve memory and protect brain tissue by reactivating this system, it would suggest that age-related cognitive decline might be more reversible than current frameworks assume.
In this view, Alzheimer’s may not stem solely from the accumulation of plaques and tangles but also from the gradual weakening of the body’s own repair systems. If that’s the case, then interventions like diAcCA that help restore lost resilience could offer benefits that are less about targeting disease directly and more about improving the brain’s ability to take care of itself.
The implications extend beyond Alzheimer’s. Nrf2 suppression is observed in other chronic conditions linked to aging, including cardiovascular disease and Parkinson’s. Whether compounds like diAcCA can temper the course of these diseases remains unanswered—but the biological overlap invites deeper exploration.
For now, the promise of diAcCA lies in the possibility that age-related brain decline is not an irreversible unraveling but rather a system-wide faltering that, with the right tools, might be slowed—or even rewired.
Model: FxFAD mice
Dosage: 10, 20, 50 mg/kg diAcCA

